MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = mol / L Calculate. - ppt download

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = mol / L Calculate. - ppt download

Chemistry Mug, 1 Mole per Liter, 1 Mole/liter, Science Teacher Gifts, Mol per Litre Coffee Cup, Funny Science Mug, Chemistry Pun - Etsy UK

Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download

PPT - MOLARITY A measurement of the concentration of a solution PowerPoint Presentation - ID:1459950

Kimia 1 Mol Per Liter untuk Mol atau Mug Teh Sederhana Perlengkapan Minum Hadiah Foto Pegangan Bulat Cangkir Dicetak Gambar Desain Kopi - AliExpress

SOLVED: Calculate the density, in grams per liter, for NOCl gas at 22°C and 608 mm Hg: Hint: Assume 1 mol of gas. 1.28 g/L 970 g/L 0.783 g/L 1.00 g/L 2.98x10^-4
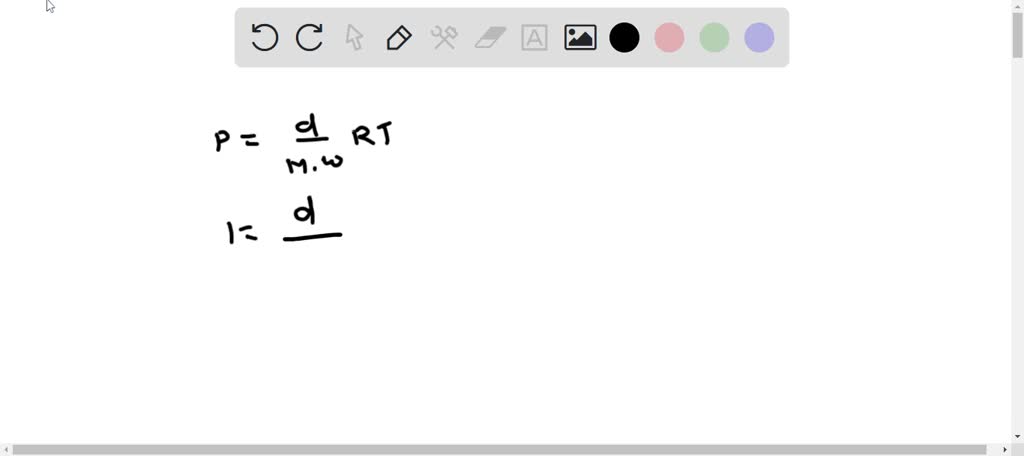
SOLVED: What is the approximate density of 1 mole of fluorine gas, F2, (molar mass = 70.1 g/mol) in units of grams per liter at STP, given that 1 mol = 22.414

PPT - MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of PowerPoint Presentation - ID:442848

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = n / V = mol / L Calculate. - ppt download